This text has been reviewed in keeping with Science X’s editorial procedure
and insurance policies.
Editors have highlighted the next attributes whilst making sure the content material’s credibility:
fact-checked
peer-reviewed e-newsletter
depended on supply
proofread
Good enough!
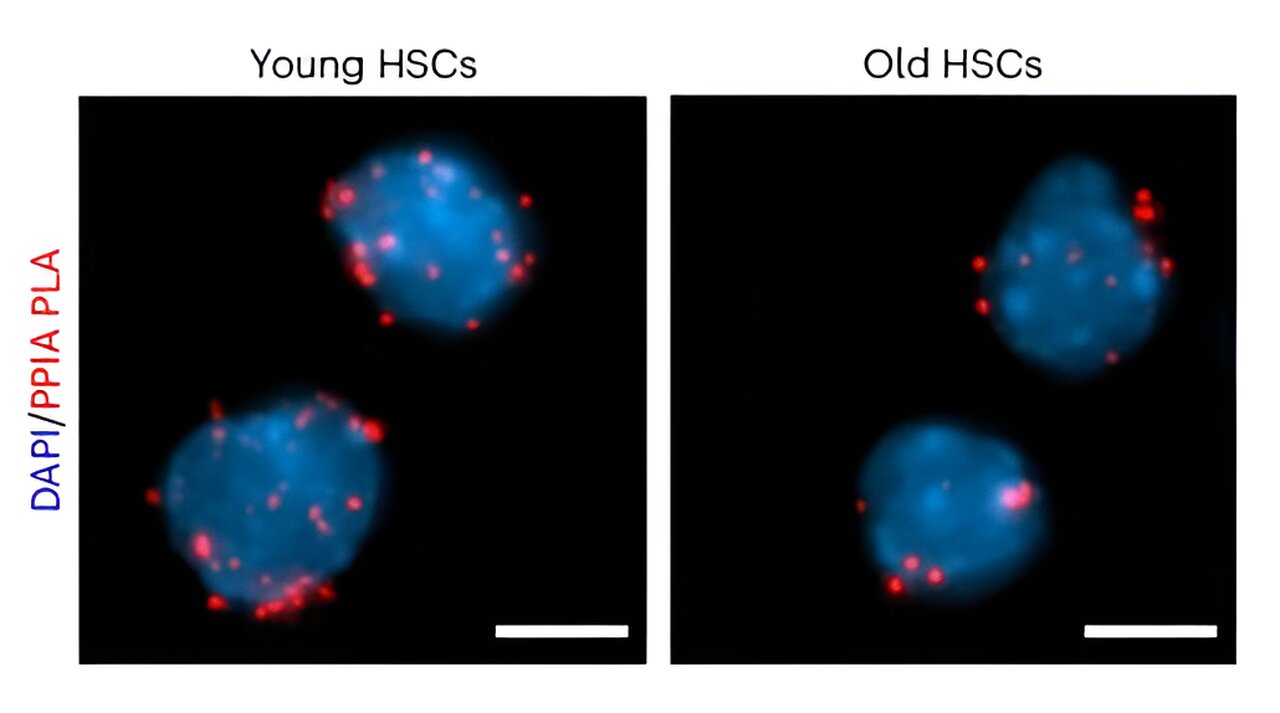
The degrees of cyclophilin A, proven right here in pink, decline as hematopoietic stem cells age. Credit score: Nature Mobile Biology (2024). DOI: 10.1038/s41556-024-01387-x
× shut
The degrees of cyclophilin A, proven right here in pink, decline as hematopoietic stem cells age. Credit score: Nature Mobile Biology (2024). DOI: 10.1038/s41556-024-01387-x
Not anything lives without end, however in comparison to different cells within the frame, hematopoietic stem cells (HSCs) are remarkably long-lived. HSCs are blood-forming cells—they provide upward thrust to abruptly dividing progenitor cells, which in flip generate masses of billions of cells to satisfy the day by day call for of oxygen-delivering pink blood cells, disease-fighting white blood cells and clot-forming platelets.
HSCs most often stay dormant inside the bone marrow, but they possess the power to turn on and fill up blood cells regularly, keeping up a slightly younger profile during the lifetime of an organism. What’s the name of the game of long-lived HSCs that wards off the consequences of growing old? A crew led via researchers at Baylor School of Medication has published in Nature Mobile Biology that the enzyme cyclophilin A, which is produced in huge quantities in HSCs, is essential for those cells to retain their regenerative doable and avert the consequences of growing old.
Lengthy reside the stem cells
“A driver of cell growing old is the buildup of proteins that experience reached the top in their helpful lifestyles,” stated corresponding writer Dr. André Catic, assistant professor and CPRIT Pupil in Most cancers Analysis within the Huffington Heart on Getting old at Baylor. “With age, proteins have a tendency to misfold, mixture and acquire throughout the cellular, which results in poisonous rigidity that may disrupt cellular serve as.”
Cells that incessantly interact in cellular department, like progenitor cells, can eliminate protein aggregates thru dilution. Alternatively, long-lived HSCs, which don’t divide incessantly, face the issue of the buildup of misfolded proteins and next poisonous rigidity. Nonetheless, HSCs stay impervious to growing old. How do they do it?
“Working out the molecular mechanisms that give a contribution to HSC growing old no longer handiest contributes to the sector of standard HSC biology, but in addition will have vital scientific relevance for most cancers remedy,” stated co-first writer of the paintings, Dr. Lauren Maneix, who was once on the Catic lab whilst operating in this mission.
Molecular chaperones at paintings
Earlier research have proven that mammalian cells specific a number of masses of molecular chaperones, proteins that maintain or alternate the 3-dimensional conformation of current proteins. Cyclophilins, one of the vital ample chaperones, had been implicated within the growing old procedure. On the other hand, how they impact cell proteins has no longer in the past been studied.
Operating with mice, the researchers first characterised the protein content material of HSCs and came upon that cyclophilin A is a prevalent chaperone. Additional experiments confirmed that the expression of cyclophilin A was once considerably diminished in elderly HSCs, and genetically getting rid of cyclophilin A speeded up herbal growing old within the stem cellular compartment. Against this, reintroducing cyclophilin A into elderly HSCs enhanced their serve as. In combination, those findings enhance cyclophilin A as a key issue within the longevity of HSCs.
Connecting cyclophilin A, intrinsically disordered proteins and HSC longevity
Subsequent, the crew investigated the proteins with which cyclophilin A interacts, conserving their steadiness. “We discovered that proteins enriched in intrinsically disordered areas are widespread objectives of the chaperone,” Catic stated.
Intrinsically disordered proteins naturally alternate their 3-D conformation to engage with other proteins, nucleic acids or different molecules. Because of this, proteins wealthy in intrinsically disordered areas control many cell processes via selling explicit actions between molecules.
“Because of their versatile nature, intrinsically disordered proteins are inherently liable to aggregation. Cyclophilin A helps those proteins in satisfying their purposes and concurrently prevents them from clumping,” Catic stated.
Moreover, the findings counsel that cyclophilin A interacts with intrinsically disordered proteins from the instant in their synthesis. “As those proteins are being made, cyclophilin A makes positive they retain the suitable conformations and are maintained at enough ranges,” Catic stated. “Genetic depletion of cyclophilin A leads to stem cells distinctively missing intrinsically disordered proteins.”
“For the primary time, our learn about confirmed that generating disordered proteins and keeping up the structural range of the proteins in a cellular performs a job in HSC growing old,” Maneix stated.
Co-first writer Polina Iakova, Charles G. Lee, Shannon E. Moree, Xuan Lu, Gandhar Ok. Datar, Cedric T. Hill, Eric Spooner, Jordon C. Ok. King, David B. Sykes, Borja Saez, Bruno Di Stefano, Xi Chen, Daniela S. Krause, Ergun Sahin, Francis T. F. Tsai, Margaret A. Goodell, Bradford C. Berk and David T. Scadden additionally contributed to this learn about.
Additional info:
Laure Maneix et al, Cyclophilin A helps translation of intrinsically disordered proteins and impacts haematopoietic stem cellular growing older, Nature Mobile Biology (2024). DOI: 10.1038/s41556-024-01387-x
Magazine knowledge:
Nature Mobile Biology